The H3Africa Biorepository Program manages Biospecimen collections from the Human Heredity and Health in Africa (H3Africa) Consortium. H3Africa studies are currently collecting DNA and other Biospecimens from participants in 22 African countries to assess genomic and environmental influences on communicable and non-communicable diseases.
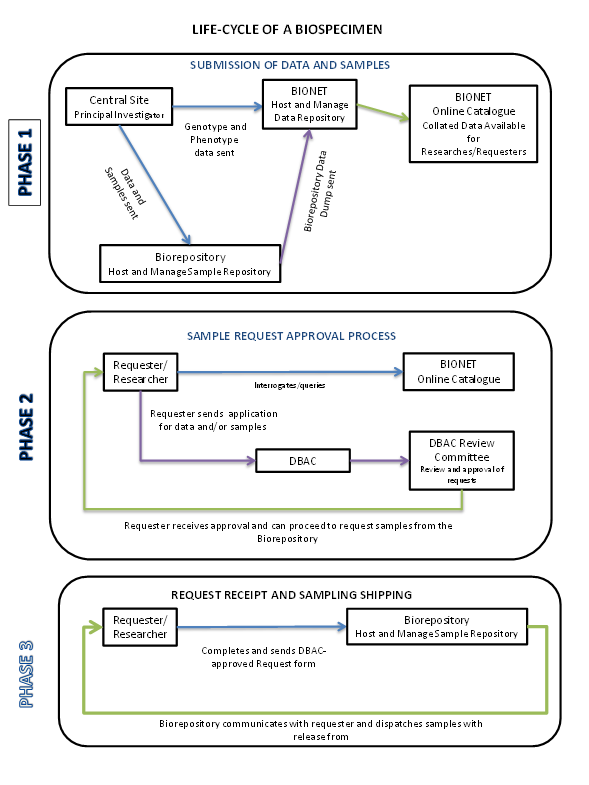
The H3Africa Initiative is committed to storing and providing samples generated by the H3Africa research projects to the wider research community. All the samples collected from the different studies are currently being deposited at the three Biorepositories in Uganda, Nigeria, and South Africa.
Initiating a Deposit
The Biorepository Program assists with, and covers costs of shipping, depositing and storing DNA from H3Africa studies, for sharing with authorized researchers. Submission of other types of Biospecimens and collections from non-H3Africa studies may also be feasible, however cost coverage is not guaranteed at this time. Each donor study is paired with a host biorepository based on geographic proximity and/or other considerations to maximize efficiency. The Biospecimens are shipped to the host biorepository from a central submission site using a courier service designated by the Biorepository. The policies and procedures for transferring the collections are described in the Biospecimen Deposit Guidelines.
The deposit process begins with planning discussions and exchange of the signed H3Africa Material Transfer Agreement, or a similar agreement, between the donor and biorepository institutions. The host biorepository should receive a copy of the study’s informed consent and ethics approval. The submission site should use the Shipment Checklist form to help prepare the shipment and email the completed checklist to the biorepository. The submission site must also submit the Shipment Manifest form to annotate the Biospecimens and minimum associated dataset. The donor investigator may also submit the optional PI Input form if there are any recommendations about future use of the collection. When a shipment is received, the host biorepository must return a Shipment Receipt and Query Form to acknowledge receipt and address any questions or concerns.
The following requirements should be met prior to deposit:-
The Biospecimens Deposit Guidelines describe the regulatory requirements, instructions, and required documentation for submitting H3Africa biospecimen collections to host H3Africa biorepositories for sharing with authorized researchers. H3Africa genomics researchers should contact their closest host H3Africa biorepository in their country or geographical region when a collection is ready for deposit. For contact information click here!
- Ethics: The Ethics Committee of the country of origin oversees the consent process and other ethical aspects related to the acquisition and subsequent use of Biospecimens, including protection of study participant (donor) privacy and confidentiality of associated data. In cases where the local Ethics Committee must approve each new use of shared Biospecimens, responsibility for obtaining approval lies with authorized users of the collection rather than the original study investigators.
- Biospecimen Deposit Material Transfer Agreement (BDMTA): The PI and signing official of the submitting institution should sign the BDMTA, which establishes the terms and conditions of Biospecimen transfer and use. The BDMTA is co-signed at the host biorepository and a signed copy is returned to the submitting institution. If required, submitting institutions may use their own MTA with similar terms and conditions, addressing the purpose of transfer, and the requirements and restrictions for Biospecimen use.
- Permits: The submitting institution and host biorepository should ensure that they have applied for, received and exchanged the relevant import and export permits for Biospecimen shipment and receipt. Copies of permits must be available at both sites.
For more information, please download the H3Africa Biospecimen Deposit Guidelines
H3Africa Biospecimen Lifecycle